Zetlam TM
Listen to the Pronunciation:
You should seek medical advice in relation to medicines and use only as directed by a healthcare professional. Always read the label. If symptoms persist see your healthcare professional.
Active ingredients: lamivudine
Brand name
(ARTG)
: ZETLAM lamivudine 100 mg tablet bottleConsumer Medicine Information (CMI)
Read the CMI leaflet for facts you need to know before, during and after taking your medicine.
Listen to the audio transcription of the CMI leaflet.
For more information about CMIs and how to read them, please visit How to read Consumer Medicine Information (CMI).
What this medicine is used for
(ARTG)
Zetlam (lamivudine) is indicated for the treatment of children (2 years and above), adolescent and adult patients with chronic hepatitis B and evidence of hepatitis B virus (HBV) replication. This indication is based on changes in serological and histological markers in clinical studies of up to 2 years duration in adult patients with compensated liver disease and serological data up to 18 months in children and adolescents. Children and adolescents also require evidence of active hepatic inflammation. (see Section 5.1 PHARMACODYNAMIC PROPERITIES, Clinical Trials). The safety and efficacy of Zetlam (lamivudine) have not been established in patients with decompensated liver disease in placebo controlled studies. However, Zetlam (lamivudine) has been shown to reduce HBV DNA levels prior to and post liver transplantation
How to use this medicine
(ARTG)
This medicine contains one component only.
Component :
- Tablet, film coated
- Oral
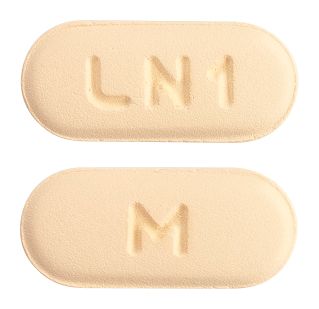
- A peach, film-coated, capsule shaped, biconvex beveled edge tablet debossed with 'LN1' on one side and 'M' on the other side.
Images

Images © Medicines Information Pty Ltd.
Storage conditions
(ARTG)
- Store below 30 degrees Celsius
- Shelf lifetime is 36 Months.
Do I need a prescription ?
(ARTG)
These medicine packs are available from a pharmacist and requires a prescription. It is
- 28 pack
- 84 pack
Is this medicine subsidised ?
(PBS)
This medicine was verified as being available on the PBS (Pharmaceutical Benefits Scheme) on May, 1 2026. To learn more about this subsidy, visit the Pharmaceutical Benefits Scheme (PBS) website.
Pregnant or planning a pregnancy ?
(AHT)
For the active ingredient lamivudine
You should seek advice from your doctor or pharmacist about taking this medicine. They can help you balance the risks and the benefits of this medicine during pregnancy.
Reporting side effects
You can help ensure medicines are safe by reporting the side effects you experience.
You can report side effects to your doctor, or directly at www.tga.gov.au/reporting-problems









