Valpam TM
You should seek medical advice in relation to medicines and use only as directed by a healthcare professional. Always read the label. If symptoms persist see your healthcare professional.
Active ingredients: diazepam
Brand name
(ARTG)
: VALPAM 2 diazepam 2 mg tablet blister packConsumer Medicine Information (CMI)
Read the CMI leaflet for facts you need to know before, during and after taking your medicine.
For more information about CMIs and how to read them, please visit How to read Consumer Medicine Information (CMI).
What this medicine is used for
(ARTG)
Management of anxiety disorders or for the short-term relief of the symptoms of anxiety. Anxiety or tension associated with the stress of everyday life usually does not require treatment with an anxiolytic. In acute alcohol withdrawal, diazepam may be useful in the symptomatic relief of acute agitation, tremor, impending or acute delirium tremens and hallucinosis. Diazepam is a useful adjunct for the relief of reflex muscle spasm due to local trauma (injury, inflammation) to muscles, bones and joints. It can also be used to combat spasticity due to upper motor neuron lesions such as cerebral palsy and paraplegia, as well as in athetosis and stiff-man syndrome.
How to use this medicine
(ARTG)
This medicine contains one component only.
Component :
- Tablet, uncoated
- Oral
- Round, flat, bevel edged, white to off-white tablet, engraved with 'DZ' break line '2' on one side and plain on the other side.
Images


Images © Medicines Information Pty Ltd.
Storage conditions
(ARTG)
- Store below 30 degrees Celsius
- Protect from Light
- Shelf lifetime is 3 Years.
Do I need a prescription ?
(ARTG)
These medicine packs are available from a pharmacist and requires a prescription. It is Schedule 4 : Prescription Only Medicine.
- 90 tablets pack
- 50 tablets pack
- 30 tablets pack
- 60 tablets pack
Is this medicine subsidised ?
(PBS)
This medicine was verified as being available on the PBS (Pharmaceutical Benefits Scheme) on April, 1 2026. To learn more about this subsidy, visit the Pharmaceutical Benefits Scheme (PBS) website.
Over 65 ?
(AHT)
This medicine contains the active ingredients:
If you are over 65 years of age, there may be specific risks and recommendations for use of this medicine. Please discuss your individual circumstances with your pharmacist, doctor or health professional. For more information read our page on medication safety for older people.
Pregnant or planning a pregnancy ?
(AHT)
For the active ingredient diazepam
You should seek advice from your doctor or pharmacist about taking this medicine. They can help you balance the risks and the benefits of this medicine during pregnancy.
Reporting side effects
You can help ensure medicines are safe by reporting the side effects you experience.
You can report side effects to your doctor, or directly at www.tga.gov.au/reporting-problems
Brand name
(ARTG)
: VALPAM 2 diazepam 2 mg tablet bottleWhat this medicine is used for
(ARTG)
Management of anxiety disorders or for the short-term relief of the symptoms of anxiety. Anxiety or tension associated with the stress of everyday life usually does not require treatment with an anxiolytic. In acute alcohol withdrawal, diazepam may be useful in the symptomatic relief of acute agitation, tremor, impending or acute delirium tremens and hallucinosis. Diazepam is a useful adjunct for the relief of reflex muscle spasm due to local trauma (injury, inflammation) to muscles, bones and joints. It can also be used to combat spasticity due to upper motor neuron lesions such as cerebral palsy and paraplegia, as well as in athetosis and stiff-man syndrome.
How to use this medicine
(ARTG)
This medicine contains one component only.
Component :
- Tablet, uncoated
- Oral
- Round, flat, bevel edged, white to off-white tablet, engraved with 'DZ' break line '2' on one side and plain on the other side.
Storage conditions
(ARTG)
- Store below 30 degrees Celsius
- Protect from Light
- Shelf lifetime is 3 Years.
Do I need a prescription ?
(ARTG)
These medicine packs are available from a pharmacist and requires a prescription. It is Schedule 4 : Prescription Only Medicine.
- 60 tablets pack
- 200 Tablets, hospital use only pack
- 30 tablets pack
- 90 tablets pack
- 50 tablets pack
- 100 Tablets, Hospital use only pack
Is this medicine subsidised ?
(PBS)
This medicine was verified as being available on the PBS (Pharmaceutical Benefits Scheme) on April, 1 2026. To learn more about this subsidy, visit the Pharmaceutical Benefits Scheme (PBS) website.
Over 65 ?
(AHT)
This medicine contains the active ingredients:
If you are over 65 years of age, there may be specific risks and recommendations for use of this medicine. Please discuss your individual circumstances with your pharmacist, doctor or health professional. For more information read our page on medication safety for older people.
Pregnant or planning a pregnancy ?
(AHT)
For the active ingredient diazepam
You should seek advice from your doctor or pharmacist about taking this medicine. They can help you balance the risks and the benefits of this medicine during pregnancy.
Reporting side effects
You can help ensure medicines are safe by reporting the side effects you experience.
You can report side effects to your doctor, or directly at www.tga.gov.au/reporting-problems
Brand name
(ARTG)
: VALPAM 5 diazepam 5 mg tablet blister packConsumer Medicine Information (CMI)
Read the CMI leaflet for facts you need to know before, during and after taking your medicine.
For more information about CMIs and how to read them, please visit How to read Consumer Medicine Information (CMI).
What this medicine is used for
(ARTG)
Management of anxiety disorders or for the short-term relief of the symptoms of anxiety. Anxiety or tension associated with the stress of everyday life usually does not require treatment with an anxiolytic. In acute alcohol withdrawal, diazepam may be useful in the symptomatic relief of acute agitation, tremor, impending or acute delirium tremens and hallucinosis. Diazepam is a useful adjunct for the relief of reflex muscle spasm due to local trauma (injury, inflammation) to muscles, bones and joints. It can also be used to combat spasticity due to upper motor neuron lesions such as cerebral palsy and paraplegia, as well as in athetosis and stiff-man syndrome.
How to use this medicine
(ARTG)
This medicine contains one component only.
Component :
- Tablet, uncoated
- Oral
- Round, flat, bevel edged, light yellow tablet, engraved with 'DZ' break line '5' on one side and plain on the other side.
Images


Images © Medicines Information Pty Ltd.
Storage conditions
(ARTG)
- Store below 30 degrees Celsius
- Protect from Light
- Shelf lifetime is 3 Years.
Do I need a prescription ?
(ARTG)
These medicine packs are available from a pharmacist and requires a prescription. It is Schedule 4 : Prescription Only Medicine.
- 60 tablets pack
- 50 tablets pack
- 90 tablets pack
- 30 tablets pack
Is this medicine subsidised ?
(PBS)
This medicine was verified as being available on the PBS (Pharmaceutical Benefits Scheme) on April, 1 2026. To learn more about this subsidy, visit the Pharmaceutical Benefits Scheme (PBS) website.
Over 65 ?
(AHT)
This medicine contains the active ingredients:
If you are over 65 years of age, there may be specific risks and recommendations for use of this medicine. Please discuss your individual circumstances with your pharmacist, doctor or health professional. For more information read our page on medication safety for older people.
Pregnant or planning a pregnancy ?
(AHT)
For the active ingredient diazepam
You should seek advice from your doctor or pharmacist about taking this medicine. They can help you balance the risks and the benefits of this medicine during pregnancy.
Reporting side effects
You can help ensure medicines are safe by reporting the side effects you experience.
You can report side effects to your doctor, or directly at www.tga.gov.au/reporting-problems
Brand name
(ARTG)
: VALPAM 5 diazepam 5 mg tablet bottleConsumer Medicine Information (CMI)
Read the CMI leaflet for facts you need to know before, during and after taking your medicine.
For more information about CMIs and how to read them, please visit How to read Consumer Medicine Information (CMI).
What this medicine is used for
(ARTG)
Management of anxiety disorders or for the short-term relief of the symptoms of anxiety. Anxiety or tension associated with the stress of everyday life usually does not require treatment with an anxiolytic. In acute alcohol withdrawal, diazepam may be useful in the symptomatic relief of acute agitation, tremor, impending or acute delirium tremens and hallucinosis. Diazepam is a useful adjunct for the relief of reflex muscle spasm due to local trauma (injury, inflammation) to muscles, bones and joints. It can also be used to combat spasticity due to upper motor neuron lesions such as cerebral palsy and paraplegia, as well as in athetosis and stiff-man syndrome.
How to use this medicine
(ARTG)
This medicine contains one component only.
Component :
- Tablet, uncoated
- Oral
- Round, flat, bevel edged, light yellow tablet, engraved with 'DZ' break line '5' on one side and plain on the other side.
Images
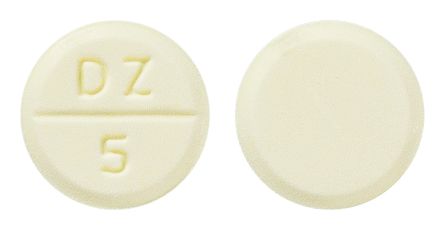

Images © Medicines Information Pty Ltd.
Storage conditions
(ARTG)
- Store below 30 degrees Celsius
- Protect from Light
- Shelf lifetime is 3 Years.
Do I need a prescription ?
(ARTG)
These medicine packs are available from a pharmacist and requires a prescription. It is Schedule 4 : Prescription Only Medicine.
- 200 Tablets, hospital use only pack
- 60 tablets pack
- 50 tablets pack
- 30 tablets pack
- 100 Tablets, hospital use only pack
- 90 tablets pack
Is this medicine subsidised ?
(PBS)
This medicine was verified as being available on the PBS (Pharmaceutical Benefits Scheme) on April, 1 2026. To learn more about this subsidy, visit the Pharmaceutical Benefits Scheme (PBS) website.
Over 65 ?
(AHT)
This medicine contains the active ingredients:
If you are over 65 years of age, there may be specific risks and recommendations for use of this medicine. Please discuss your individual circumstances with your pharmacist, doctor or health professional. For more information read our page on medication safety for older people.
Pregnant or planning a pregnancy ?
(AHT)
For the active ingredient diazepam
You should seek advice from your doctor or pharmacist about taking this medicine. They can help you balance the risks and the benefits of this medicine during pregnancy.
Reporting side effects
You can help ensure medicines are safe by reporting the side effects you experience.
You can report side effects to your doctor, or directly at www.tga.gov.au/reporting-problems
Brand name
(ARTG)
: VALPAM 2 diazepam 2 mg tablet bottleWhat this medicine is used for
(ARTG)
Management of anxiety disorders or for the short-term relief of the symptoms of anxiety. Anxiety or tension associated with the stress of everyday life usually does not require treatment with an anxiolytic. In acute alcohol withdrawal, diazepam may be useful in the symptomatic relief of acute agitation, tremor, impending or acute delirium tremens and hallucinosis. Diazepam is a useful adjunct for the relief of reflex muscle spasm due to local trauma (injury, inflammation) to muscles, bones and joints. It can also be used to combat spasticity due to upper motor neuron lesions such as cerebral palsy and paraplegia, as well as in athetosis and stiff-man syndrome.
How to use this medicine
(ARTG)
This medicine contains one component only.
Component :
- Tablet, uncoated
- Oral
- Round, flat, bevel edged, white to off-white tablet, engraved with 'DZ' break line '2' on one side and plain on the other side.
Storage conditions
(ARTG)
- Store below 30 degrees Celsius
- Protect from Light
- Shelf lifetime is 3 Years.
Do I need a prescription ?
(ARTG)
These medicine packs are available from a pharmacist and requires a prescription. It is Schedule 4 : Prescription Only Medicine.
- 60 tablets pack
- 200 Tablets, hospital use only pack
- 30 tablets pack
- 90 tablets pack
- 50 tablets pack
- 100 Tablets, Hospital use only pack
Over 65 ?
(AHT)
This medicine contains the active ingredients:
If you are over 65 years of age, there may be specific risks and recommendations for use of this medicine. Please discuss your individual circumstances with your pharmacist, doctor or health professional. For more information read our page on medication safety for older people.
Pregnant or planning a pregnancy ?
(AHT)
For the active ingredient diazepam
You should seek advice from your doctor or pharmacist about taking this medicine. They can help you balance the risks and the benefits of this medicine during pregnancy.
Reporting side effects
You can help ensure medicines are safe by reporting the side effects you experience.
You can report side effects to your doctor, or directly at www.tga.gov.au/reporting-problems
Brand name
(ARTG)
: VALPAM 2 diazepam 2 mg tablet bottleWhat this medicine is used for
(ARTG)
Management of anxiety disorders or for the short-term relief of the symptoms of anxiety. Anxiety or tension associated with the stress of everyday life usually does not require treatment with an anxiolytic. In acute alcohol withdrawal, diazepam may be useful in the symptomatic relief of acute agitation, tremor, impending or acute delirium tremens and hallucinosis. Diazepam is a useful adjunct for the relief of reflex muscle spasm due to local trauma (injury, inflammation) to muscles, bones and joints. It can also be used to combat spasticity due to upper motor neuron lesions such as cerebral palsy and paraplegia, as well as in athetosis and stiff-man syndrome.
How to use this medicine
(ARTG)
This medicine contains one component only.
Component :
- Tablet, uncoated
- Oral
- Round, flat, bevel edged, white to off-white tablet, engraved with 'DZ' break line '2' on one side and plain on the other side.
Storage conditions
(ARTG)
- Store below 30 degrees Celsius
- Protect from Light
- Shelf lifetime is 3 Years.
Do I need a prescription ?
(ARTG)
These medicine packs are available from a pharmacist and requires a prescription. It is Schedule 4 : Prescription Only Medicine.
- 60 tablets pack
- 200 Tablets, hospital use only pack
- 30 tablets pack
- 90 tablets pack
- 50 tablets pack
- 100 Tablets, Hospital use only pack
Over 65 ?
(AHT)
This medicine contains the active ingredients:
If you are over 65 years of age, there may be specific risks and recommendations for use of this medicine. Please discuss your individual circumstances with your pharmacist, doctor or health professional. For more information read our page on medication safety for older people.
Pregnant or planning a pregnancy ?
(AHT)
For the active ingredient diazepam
You should seek advice from your doctor or pharmacist about taking this medicine. They can help you balance the risks and the benefits of this medicine during pregnancy.
Reporting side effects
You can help ensure medicines are safe by reporting the side effects you experience.
You can report side effects to your doctor, or directly at www.tga.gov.au/reporting-problems
Brand name
(ARTG)
: VALPAM 2 diazepam 2 mg tablet bottleWhat this medicine is used for
(ARTG)
Management of anxiety disorders or for the short-term relief of the symptoms of anxiety. Anxiety or tension associated with the stress of everyday life usually does not require treatment with an anxiolytic. In acute alcohol withdrawal, diazepam may be useful in the symptomatic relief of acute agitation, tremor, impending or acute delirium tremens and hallucinosis. Diazepam is a useful adjunct for the relief of reflex muscle spasm due to local trauma (injury, inflammation) to muscles, bones and joints. It can also be used to combat spasticity due to upper motor neuron lesions such as cerebral palsy and paraplegia, as well as in athetosis and stiff-man syndrome.
How to use this medicine
(ARTG)
This medicine contains one component only.
Component :
- Tablet, uncoated
- Oral
- Round, flat, bevel edged, white to off-white tablet, engraved with 'DZ' break line '2' on one side and plain on the other side.
Storage conditions
(ARTG)
- Store below 30 degrees Celsius
- Protect from Light
- Shelf lifetime is 3 Years.
Do I need a prescription ?
(ARTG)
These medicine packs are available from a pharmacist and requires a prescription. It is Schedule 4 : Prescription Only Medicine.
- 60 tablets pack
- 200 Tablets, hospital use only pack
- 30 tablets pack
- 90 tablets pack
- 50 tablets pack
- 100 Tablets, Hospital use only pack
Over 65 ?
(AHT)
This medicine contains the active ingredients:
If you are over 65 years of age, there may be specific risks and recommendations for use of this medicine. Please discuss your individual circumstances with your pharmacist, doctor or health professional. For more information read our page on medication safety for older people.
Pregnant or planning a pregnancy ?
(AHT)
For the active ingredient diazepam
You should seek advice from your doctor or pharmacist about taking this medicine. They can help you balance the risks and the benefits of this medicine during pregnancy.
Reporting side effects
You can help ensure medicines are safe by reporting the side effects you experience.
You can report side effects to your doctor, or directly at www.tga.gov.au/reporting-problems
Brand name
(ARTG)
: VALPAM 2 diazepam 2 mg tablet bottleWhat this medicine is used for
(ARTG)
Management of anxiety disorders or for the short-term relief of the symptoms of anxiety. Anxiety or tension associated with the stress of everyday life usually does not require treatment with an anxiolytic. In acute alcohol withdrawal, diazepam may be useful in the symptomatic relief of acute agitation, tremor, impending or acute delirium tremens and hallucinosis. Diazepam is a useful adjunct for the relief of reflex muscle spasm due to local trauma (injury, inflammation) to muscles, bones and joints. It can also be used to combat spasticity due to upper motor neuron lesions such as cerebral palsy and paraplegia, as well as in athetosis and stiff-man syndrome.
How to use this medicine
(ARTG)
This medicine contains one component only.
Component :
- Tablet, uncoated
- Oral
- Round, flat, bevel edged, white to off-white tablet, engraved with 'DZ' break line '2' on one side and plain on the other side.
Storage conditions
(ARTG)
- Store below 30 degrees Celsius
- Protect from Light
- Shelf lifetime is 3 Years.
Do I need a prescription ?
(ARTG)
These medicine packs are available from a pharmacist and requires a prescription. It is Schedule 4 : Prescription Only Medicine.
- 60 tablets pack
- 200 Tablets, hospital use only pack
- 30 tablets pack
- 90 tablets pack
- 50 tablets pack
- 100 Tablets, Hospital use only pack
Over 65 ?
(AHT)
This medicine contains the active ingredients:
If you are over 65 years of age, there may be specific risks and recommendations for use of this medicine. Please discuss your individual circumstances with your pharmacist, doctor or health professional. For more information read our page on medication safety for older people.
Pregnant or planning a pregnancy ?
(AHT)
For the active ingredient diazepam
You should seek advice from your doctor or pharmacist about taking this medicine. They can help you balance the risks and the benefits of this medicine during pregnancy.
Reporting side effects
You can help ensure medicines are safe by reporting the side effects you experience.
You can report side effects to your doctor, or directly at www.tga.gov.au/reporting-problems
Brand name
(ARTG)
: VALPAM 2 diazepam 2 mg tablet bottleWhat this medicine is used for
(ARTG)
Management of anxiety disorders or for the short-term relief of the symptoms of anxiety. Anxiety or tension associated with the stress of everyday life usually does not require treatment with an anxiolytic. In acute alcohol withdrawal, diazepam may be useful in the symptomatic relief of acute agitation, tremor, impending or acute delirium tremens and hallucinosis. Diazepam is a useful adjunct for the relief of reflex muscle spasm due to local trauma (injury, inflammation) to muscles, bones and joints. It can also be used to combat spasticity due to upper motor neuron lesions such as cerebral palsy and paraplegia, as well as in athetosis and stiff-man syndrome.
How to use this medicine
(ARTG)
This medicine contains one component only.
Component :
- Tablet, uncoated
- Oral
- Round, flat, bevel edged, white to off-white tablet, engraved with 'DZ' break line '2' on one side and plain on the other side.
Storage conditions
(ARTG)
- Store below 30 degrees Celsius
- Protect from Light
- Shelf lifetime is 3 Years.
Do I need a prescription ?
(ARTG)
These medicine packs are available from a pharmacist and requires a prescription. It is Schedule 4 : Prescription Only Medicine.
- 60 tablets pack
- 200 Tablets, hospital use only pack
- 30 tablets pack
- 90 tablets pack
- 50 tablets pack
- 100 Tablets, Hospital use only pack
Over 65 ?
(AHT)
This medicine contains the active ingredients:
If you are over 65 years of age, there may be specific risks and recommendations for use of this medicine. Please discuss your individual circumstances with your pharmacist, doctor or health professional. For more information read our page on medication safety for older people.
Pregnant or planning a pregnancy ?
(AHT)
For the active ingredient diazepam
You should seek advice from your doctor or pharmacist about taking this medicine. They can help you balance the risks and the benefits of this medicine during pregnancy.
Reporting side effects
You can help ensure medicines are safe by reporting the side effects you experience.
You can report side effects to your doctor, or directly at www.tga.gov.au/reporting-problems
Brand name
(ARTG)
: VALPAM 2 diazepam 2 mg tablet blister packConsumer Medicine Information (CMI)
Read the CMI leaflet for facts you need to know before, during and after taking your medicine.
For more information about CMIs and how to read them, please visit How to read Consumer Medicine Information (CMI).
What this medicine is used for
(ARTG)
Management of anxiety disorders or for the short-term relief of the symptoms of anxiety. Anxiety or tension associated with the stress of everyday life usually does not require treatment with an anxiolytic. In acute alcohol withdrawal, diazepam may be useful in the symptomatic relief of acute agitation, tremor, impending or acute delirium tremens and hallucinosis. Diazepam is a useful adjunct for the relief of reflex muscle spasm due to local trauma (injury, inflammation) to muscles, bones and joints. It can also be used to combat spasticity due to upper motor neuron lesions such as cerebral palsy and paraplegia, as well as in athetosis and stiff-man syndrome.
How to use this medicine
(ARTG)
This medicine contains one component only.
Component :
- Tablet, uncoated
- Oral
- Round, flat, bevel edged, white to off-white tablet, engraved with 'DZ' break line '2' on one side and plain on the other side.
Images


Images © Medicines Information Pty Ltd.
Storage conditions
(ARTG)
- Store below 30 degrees Celsius
- Protect from Light
- Shelf lifetime is 3 Years.
Do I need a prescription ?
(ARTG)
These medicine packs are available from a pharmacist and requires a prescription. It is Schedule 4 : Prescription Only Medicine.
- 90 tablets pack
- 50 tablets pack
- 30 tablets pack
- 60 tablets pack
Over 65 ?
(AHT)
This medicine contains the active ingredients:
If you are over 65 years of age, there may be specific risks and recommendations for use of this medicine. Please discuss your individual circumstances with your pharmacist, doctor or health professional. For more information read our page on medication safety for older people.
Pregnant or planning a pregnancy ?
(AHT)
For the active ingredient diazepam
You should seek advice from your doctor or pharmacist about taking this medicine. They can help you balance the risks and the benefits of this medicine during pregnancy.
Reporting side effects
You can help ensure medicines are safe by reporting the side effects you experience.
You can report side effects to your doctor, or directly at www.tga.gov.au/reporting-problems
Brand name
(ARTG)
: VALPAM 2 diazepam 2 mg tablet blister packConsumer Medicine Information (CMI)
Read the CMI leaflet for facts you need to know before, during and after taking your medicine.
For more information about CMIs and how to read them, please visit How to read Consumer Medicine Information (CMI).
What this medicine is used for
(ARTG)
Management of anxiety disorders or for the short-term relief of the symptoms of anxiety. Anxiety or tension associated with the stress of everyday life usually does not require treatment with an anxiolytic. In acute alcohol withdrawal, diazepam may be useful in the symptomatic relief of acute agitation, tremor, impending or acute delirium tremens and hallucinosis. Diazepam is a useful adjunct for the relief of reflex muscle spasm due to local trauma (injury, inflammation) to muscles, bones and joints. It can also be used to combat spasticity due to upper motor neuron lesions such as cerebral palsy and paraplegia, as well as in athetosis and stiff-man syndrome.
How to use this medicine
(ARTG)
This medicine contains one component only.
Component :
- Tablet, uncoated
- Oral
- Round, flat, bevel edged, white to off-white tablet, engraved with 'DZ' break line '2' on one side and plain on the other side.
Images


Images © Medicines Information Pty Ltd.
Storage conditions
(ARTG)
- Store below 30 degrees Celsius
- Protect from Light
- Shelf lifetime is 3 Years.
Do I need a prescription ?
(ARTG)
These medicine packs are available from a pharmacist and requires a prescription. It is Schedule 4 : Prescription Only Medicine.
- 90 tablets pack
- 50 tablets pack
- 30 tablets pack
- 60 tablets pack
Over 65 ?
(AHT)
This medicine contains the active ingredients:
If you are over 65 years of age, there may be specific risks and recommendations for use of this medicine. Please discuss your individual circumstances with your pharmacist, doctor or health professional. For more information read our page on medication safety for older people.
Pregnant or planning a pregnancy ?
(AHT)
For the active ingredient diazepam
You should seek advice from your doctor or pharmacist about taking this medicine. They can help you balance the risks and the benefits of this medicine during pregnancy.
Reporting side effects
You can help ensure medicines are safe by reporting the side effects you experience.
You can report side effects to your doctor, or directly at www.tga.gov.au/reporting-problems
Brand name
(ARTG)
: VALPAM 2 diazepam 2 mg tablet blister packConsumer Medicine Information (CMI)
Read the CMI leaflet for facts you need to know before, during and after taking your medicine.
For more information about CMIs and how to read them, please visit How to read Consumer Medicine Information (CMI).
What this medicine is used for
(ARTG)
Management of anxiety disorders or for the short-term relief of the symptoms of anxiety. Anxiety or tension associated with the stress of everyday life usually does not require treatment with an anxiolytic. In acute alcohol withdrawal, diazepam may be useful in the symptomatic relief of acute agitation, tremor, impending or acute delirium tremens and hallucinosis. Diazepam is a useful adjunct for the relief of reflex muscle spasm due to local trauma (injury, inflammation) to muscles, bones and joints. It can also be used to combat spasticity due to upper motor neuron lesions such as cerebral palsy and paraplegia, as well as in athetosis and stiff-man syndrome.
How to use this medicine
(ARTG)
This medicine contains one component only.
Component :
- Tablet, uncoated
- Oral
- Round, flat, bevel edged, white to off-white tablet, engraved with 'DZ' break line '2' on one side and plain on the other side.
Images


Images © Medicines Information Pty Ltd.
Storage conditions
(ARTG)
- Store below 30 degrees Celsius
- Protect from Light
- Shelf lifetime is 3 Years.
Do I need a prescription ?
(ARTG)
These medicine packs are available from a pharmacist and requires a prescription. It is Schedule 4 : Prescription Only Medicine.
- 90 tablets pack
- 50 tablets pack
- 30 tablets pack
- 60 tablets pack
Over 65 ?
(AHT)
This medicine contains the active ingredients:
If you are over 65 years of age, there may be specific risks and recommendations for use of this medicine. Please discuss your individual circumstances with your pharmacist, doctor or health professional. For more information read our page on medication safety for older people.
Pregnant or planning a pregnancy ?
(AHT)
For the active ingredient diazepam
You should seek advice from your doctor or pharmacist about taking this medicine. They can help you balance the risks and the benefits of this medicine during pregnancy.
Reporting side effects
You can help ensure medicines are safe by reporting the side effects you experience.
You can report side effects to your doctor, or directly at www.tga.gov.au/reporting-problems
Brand name
(ARTG)
: VALPAM 5 diazepam 5 mg tablet blister packConsumer Medicine Information (CMI)
Read the CMI leaflet for facts you need to know before, during and after taking your medicine.
For more information about CMIs and how to read them, please visit How to read Consumer Medicine Information (CMI).
What this medicine is used for
(ARTG)
Management of anxiety disorders or for the short-term relief of the symptoms of anxiety. Anxiety or tension associated with the stress of everyday life usually does not require treatment with an anxiolytic. In acute alcohol withdrawal, diazepam may be useful in the symptomatic relief of acute agitation, tremor, impending or acute delirium tremens and hallucinosis. Diazepam is a useful adjunct for the relief of reflex muscle spasm due to local trauma (injury, inflammation) to muscles, bones and joints. It can also be used to combat spasticity due to upper motor neuron lesions such as cerebral palsy and paraplegia, as well as in athetosis and stiff-man syndrome.
How to use this medicine
(ARTG)
This medicine contains one component only.
Component :
- Tablet, uncoated
- Oral
- Round, flat, bevel edged, light yellow tablet, engraved with 'DZ' break line '5' on one side and plain on the other side.
Images


Images © Medicines Information Pty Ltd.
Storage conditions
(ARTG)
- Store below 30 degrees Celsius
- Protect from Light
- Shelf lifetime is 3 Years.
Do I need a prescription ?
(ARTG)
These medicine packs are available from a pharmacist and requires a prescription. It is Schedule 4 : Prescription Only Medicine.
- 60 tablets pack
- 50 tablets pack
- 90 tablets pack
- 30 tablets pack
Over 65 ?
(AHT)
This medicine contains the active ingredients:
If you are over 65 years of age, there may be specific risks and recommendations for use of this medicine. Please discuss your individual circumstances with your pharmacist, doctor or health professional. For more information read our page on medication safety for older people.
Pregnant or planning a pregnancy ?
(AHT)
For the active ingredient diazepam
You should seek advice from your doctor or pharmacist about taking this medicine. They can help you balance the risks and the benefits of this medicine during pregnancy.
Reporting side effects
You can help ensure medicines are safe by reporting the side effects you experience.
You can report side effects to your doctor, or directly at www.tga.gov.au/reporting-problems
Brand name
(ARTG)
: VALPAM 5 diazepam 5 mg tablet blister packConsumer Medicine Information (CMI)
Read the CMI leaflet for facts you need to know before, during and after taking your medicine.
For more information about CMIs and how to read them, please visit How to read Consumer Medicine Information (CMI).
What this medicine is used for
(ARTG)
Management of anxiety disorders or for the short-term relief of the symptoms of anxiety. Anxiety or tension associated with the stress of everyday life usually does not require treatment with an anxiolytic. In acute alcohol withdrawal, diazepam may be useful in the symptomatic relief of acute agitation, tremor, impending or acute delirium tremens and hallucinosis. Diazepam is a useful adjunct for the relief of reflex muscle spasm due to local trauma (injury, inflammation) to muscles, bones and joints. It can also be used to combat spasticity due to upper motor neuron lesions such as cerebral palsy and paraplegia, as well as in athetosis and stiff-man syndrome.
How to use this medicine
(ARTG)
This medicine contains one component only.
Component :
- Tablet, uncoated
- Oral
- Round, flat, bevel edged, light yellow tablet, engraved with 'DZ' break line '5' on one side and plain on the other side.
Images


Images © Medicines Information Pty Ltd.
Storage conditions
(ARTG)
- Store below 30 degrees Celsius
- Protect from Light
- Shelf lifetime is 3 Years.
Do I need a prescription ?
(ARTG)
These medicine packs are available from a pharmacist and requires a prescription. It is Schedule 4 : Prescription Only Medicine.
- 60 tablets pack
- 50 tablets pack
- 90 tablets pack
- 30 tablets pack
Over 65 ?
(AHT)
This medicine contains the active ingredients:
If you are over 65 years of age, there may be specific risks and recommendations for use of this medicine. Please discuss your individual circumstances with your pharmacist, doctor or health professional. For more information read our page on medication safety for older people.
Pregnant or planning a pregnancy ?
(AHT)
For the active ingredient diazepam
You should seek advice from your doctor or pharmacist about taking this medicine. They can help you balance the risks and the benefits of this medicine during pregnancy.
Reporting side effects
You can help ensure medicines are safe by reporting the side effects you experience.
You can report side effects to your doctor, or directly at www.tga.gov.au/reporting-problems
Brand name
(ARTG)
: VALPAM 5 diazepam 5 mg tablet blister packConsumer Medicine Information (CMI)
Read the CMI leaflet for facts you need to know before, during and after taking your medicine.
For more information about CMIs and how to read them, please visit How to read Consumer Medicine Information (CMI).
What this medicine is used for
(ARTG)
Management of anxiety disorders or for the short-term relief of the symptoms of anxiety. Anxiety or tension associated with the stress of everyday life usually does not require treatment with an anxiolytic. In acute alcohol withdrawal, diazepam may be useful in the symptomatic relief of acute agitation, tremor, impending or acute delirium tremens and hallucinosis. Diazepam is a useful adjunct for the relief of reflex muscle spasm due to local trauma (injury, inflammation) to muscles, bones and joints. It can also be used to combat spasticity due to upper motor neuron lesions such as cerebral palsy and paraplegia, as well as in athetosis and stiff-man syndrome.
How to use this medicine
(ARTG)
This medicine contains one component only.
Component :
- Tablet, uncoated
- Oral
- Round, flat, bevel edged, light yellow tablet, engraved with 'DZ' break line '5' on one side and plain on the other side.
Images


Images © Medicines Information Pty Ltd.
Storage conditions
(ARTG)
- Store below 30 degrees Celsius
- Protect from Light
- Shelf lifetime is 3 Years.
Do I need a prescription ?
(ARTG)
These medicine packs are available from a pharmacist and requires a prescription. It is Schedule 4 : Prescription Only Medicine.
- 60 tablets pack
- 50 tablets pack
- 90 tablets pack
- 30 tablets pack
Over 65 ?
(AHT)
This medicine contains the active ingredients:
If you are over 65 years of age, there may be specific risks and recommendations for use of this medicine. Please discuss your individual circumstances with your pharmacist, doctor or health professional. For more information read our page on medication safety for older people.
Pregnant or planning a pregnancy ?
(AHT)
For the active ingredient diazepam
You should seek advice from your doctor or pharmacist about taking this medicine. They can help you balance the risks and the benefits of this medicine during pregnancy.
Reporting side effects
You can help ensure medicines are safe by reporting the side effects you experience.
You can report side effects to your doctor, or directly at www.tga.gov.au/reporting-problems
Brand name
(ARTG)
: VALPAM 5 diazepam 5 mg tablet bottleConsumer Medicine Information (CMI)
Read the CMI leaflet for facts you need to know before, during and after taking your medicine.
For more information about CMIs and how to read them, please visit How to read Consumer Medicine Information (CMI).
What this medicine is used for
(ARTG)
Management of anxiety disorders or for the short-term relief of the symptoms of anxiety. Anxiety or tension associated with the stress of everyday life usually does not require treatment with an anxiolytic. In acute alcohol withdrawal, diazepam may be useful in the symptomatic relief of acute agitation, tremor, impending or acute delirium tremens and hallucinosis. Diazepam is a useful adjunct for the relief of reflex muscle spasm due to local trauma (injury, inflammation) to muscles, bones and joints. It can also be used to combat spasticity due to upper motor neuron lesions such as cerebral palsy and paraplegia, as well as in athetosis and stiff-man syndrome.
How to use this medicine
(ARTG)
This medicine contains one component only.
Component :
- Tablet, uncoated
- Oral
- Round, flat, bevel edged, light yellow tablet, engraved with 'DZ' break line '5' on one side and plain on the other side.
Images


Images © Medicines Information Pty Ltd.
Storage conditions
(ARTG)
- Store below 30 degrees Celsius
- Protect from Light
- Shelf lifetime is 3 Years.
Do I need a prescription ?
(ARTG)
These medicine packs are available from a pharmacist and requires a prescription. It is Schedule 4 : Prescription Only Medicine.
- 200 Tablets, hospital use only pack
- 60 tablets pack
- 50 tablets pack
- 30 tablets pack
- 100 Tablets, hospital use only pack
- 90 tablets pack
Over 65 ?
(AHT)
This medicine contains the active ingredients:
If you are over 65 years of age, there may be specific risks and recommendations for use of this medicine. Please discuss your individual circumstances with your pharmacist, doctor or health professional. For more information read our page on medication safety for older people.
Pregnant or planning a pregnancy ?
(AHT)
For the active ingredient diazepam
You should seek advice from your doctor or pharmacist about taking this medicine. They can help you balance the risks and the benefits of this medicine during pregnancy.
Reporting side effects
You can help ensure medicines are safe by reporting the side effects you experience.
You can report side effects to your doctor, or directly at www.tga.gov.au/reporting-problems
Brand name
(ARTG)
: VALPAM 5 diazepam 5 mg tablet bottleConsumer Medicine Information (CMI)
Read the CMI leaflet for facts you need to know before, during and after taking your medicine.
For more information about CMIs and how to read them, please visit How to read Consumer Medicine Information (CMI).
What this medicine is used for
(ARTG)
Management of anxiety disorders or for the short-term relief of the symptoms of anxiety. Anxiety or tension associated with the stress of everyday life usually does not require treatment with an anxiolytic. In acute alcohol withdrawal, diazepam may be useful in the symptomatic relief of acute agitation, tremor, impending or acute delirium tremens and hallucinosis. Diazepam is a useful adjunct for the relief of reflex muscle spasm due to local trauma (injury, inflammation) to muscles, bones and joints. It can also be used to combat spasticity due to upper motor neuron lesions such as cerebral palsy and paraplegia, as well as in athetosis and stiff-man syndrome.
How to use this medicine
(ARTG)
This medicine contains one component only.
Component :
- Tablet, uncoated
- Oral
- Round, flat, bevel edged, light yellow tablet, engraved with 'DZ' break line '5' on one side and plain on the other side.
Images


Images © Medicines Information Pty Ltd.
Storage conditions
(ARTG)
- Store below 30 degrees Celsius
- Protect from Light
- Shelf lifetime is 3 Years.
Do I need a prescription ?
(ARTG)
These medicine packs are available from a pharmacist and requires a prescription. It is Schedule 4 : Prescription Only Medicine.
- 200 Tablets, hospital use only pack
- 60 tablets pack
- 50 tablets pack
- 30 tablets pack
- 100 Tablets, hospital use only pack
- 90 tablets pack
Over 65 ?
(AHT)
This medicine contains the active ingredients:
If you are over 65 years of age, there may be specific risks and recommendations for use of this medicine. Please discuss your individual circumstances with your pharmacist, doctor or health professional. For more information read our page on medication safety for older people.
Pregnant or planning a pregnancy ?
(AHT)
For the active ingredient diazepam
You should seek advice from your doctor or pharmacist about taking this medicine. They can help you balance the risks and the benefits of this medicine during pregnancy.
Reporting side effects
You can help ensure medicines are safe by reporting the side effects you experience.
You can report side effects to your doctor, or directly at www.tga.gov.au/reporting-problems
Brand name
(ARTG)
: VALPAM 5 diazepam 5 mg tablet bottleConsumer Medicine Information (CMI)
Read the CMI leaflet for facts you need to know before, during and after taking your medicine.
For more information about CMIs and how to read them, please visit How to read Consumer Medicine Information (CMI).
What this medicine is used for
(ARTG)
Management of anxiety disorders or for the short-term relief of the symptoms of anxiety. Anxiety or tension associated with the stress of everyday life usually does not require treatment with an anxiolytic. In acute alcohol withdrawal, diazepam may be useful in the symptomatic relief of acute agitation, tremor, impending or acute delirium tremens and hallucinosis. Diazepam is a useful adjunct for the relief of reflex muscle spasm due to local trauma (injury, inflammation) to muscles, bones and joints. It can also be used to combat spasticity due to upper motor neuron lesions such as cerebral palsy and paraplegia, as well as in athetosis and stiff-man syndrome.
How to use this medicine
(ARTG)
This medicine contains one component only.
Component :
- Tablet, uncoated
- Oral
- Round, flat, bevel edged, light yellow tablet, engraved with 'DZ' break line '5' on one side and plain on the other side.
Images


Images © Medicines Information Pty Ltd.
Storage conditions
(ARTG)
- Store below 30 degrees Celsius
- Protect from Light
- Shelf lifetime is 3 Years.
Do I need a prescription ?
(ARTG)
These medicine packs are available from a pharmacist and requires a prescription. It is Schedule 4 : Prescription Only Medicine.
- 200 Tablets, hospital use only pack
- 60 tablets pack
- 50 tablets pack
- 30 tablets pack
- 100 Tablets, hospital use only pack
- 90 tablets pack
Over 65 ?
(AHT)
This medicine contains the active ingredients:
If you are over 65 years of age, there may be specific risks and recommendations for use of this medicine. Please discuss your individual circumstances with your pharmacist, doctor or health professional. For more information read our page on medication safety for older people.
Pregnant or planning a pregnancy ?
(AHT)
For the active ingredient diazepam
You should seek advice from your doctor or pharmacist about taking this medicine. They can help you balance the risks and the benefits of this medicine during pregnancy.
Reporting side effects
You can help ensure medicines are safe by reporting the side effects you experience.
You can report side effects to your doctor, or directly at www.tga.gov.au/reporting-problems
Brand name
(ARTG)
: VALPAM 5 diazepam 5 mg tablet bottleConsumer Medicine Information (CMI)
Read the CMI leaflet for facts you need to know before, during and after taking your medicine.
For more information about CMIs and how to read them, please visit How to read Consumer Medicine Information (CMI).
What this medicine is used for
(ARTG)
Management of anxiety disorders or for the short-term relief of the symptoms of anxiety. Anxiety or tension associated with the stress of everyday life usually does not require treatment with an anxiolytic. In acute alcohol withdrawal, diazepam may be useful in the symptomatic relief of acute agitation, tremor, impending or acute delirium tremens and hallucinosis. Diazepam is a useful adjunct for the relief of reflex muscle spasm due to local trauma (injury, inflammation) to muscles, bones and joints. It can also be used to combat spasticity due to upper motor neuron lesions such as cerebral palsy and paraplegia, as well as in athetosis and stiff-man syndrome.
How to use this medicine
(ARTG)
This medicine contains one component only.
Component :
- Tablet, uncoated
- Oral
- Round, flat, bevel edged, light yellow tablet, engraved with 'DZ' break line '5' on one side and plain on the other side.
Images


Images © Medicines Information Pty Ltd.
Storage conditions
(ARTG)
- Store below 30 degrees Celsius
- Protect from Light
- Shelf lifetime is 3 Years.
Do I need a prescription ?
(ARTG)
These medicine packs are available from a pharmacist and requires a prescription. It is Schedule 4 : Prescription Only Medicine.
- 200 Tablets, hospital use only pack
- 60 tablets pack
- 50 tablets pack
- 30 tablets pack
- 100 Tablets, hospital use only pack
- 90 tablets pack
Over 65 ?
(AHT)
This medicine contains the active ingredients:
If you are over 65 years of age, there may be specific risks and recommendations for use of this medicine. Please discuss your individual circumstances with your pharmacist, doctor or health professional. For more information read our page on medication safety for older people.
Pregnant or planning a pregnancy ?
(AHT)
For the active ingredient diazepam
You should seek advice from your doctor or pharmacist about taking this medicine. They can help you balance the risks and the benefits of this medicine during pregnancy.
Reporting side effects
You can help ensure medicines are safe by reporting the side effects you experience.
You can report side effects to your doctor, or directly at www.tga.gov.au/reporting-problems
Brand name
(ARTG)
: VALPAM 5 diazepam 5 mg tablet bottleConsumer Medicine Information (CMI)
Read the CMI leaflet for facts you need to know before, during and after taking your medicine.
For more information about CMIs and how to read them, please visit How to read Consumer Medicine Information (CMI).
What this medicine is used for
(ARTG)
Management of anxiety disorders or for the short-term relief of the symptoms of anxiety. Anxiety or tension associated with the stress of everyday life usually does not require treatment with an anxiolytic. In acute alcohol withdrawal, diazepam may be useful in the symptomatic relief of acute agitation, tremor, impending or acute delirium tremens and hallucinosis. Diazepam is a useful adjunct for the relief of reflex muscle spasm due to local trauma (injury, inflammation) to muscles, bones and joints. It can also be used to combat spasticity due to upper motor neuron lesions such as cerebral palsy and paraplegia, as well as in athetosis and stiff-man syndrome.
How to use this medicine
(ARTG)
This medicine contains one component only.
Component :
- Tablet, uncoated
- Oral
- Round, flat, bevel edged, light yellow tablet, engraved with 'DZ' break line '5' on one side and plain on the other side.
Images


Images © Medicines Information Pty Ltd.
Storage conditions
(ARTG)
- Store below 30 degrees Celsius
- Protect from Light
- Shelf lifetime is 3 Years.
Do I need a prescription ?
(ARTG)
These medicine packs are available from a pharmacist and requires a prescription. It is Schedule 4 : Prescription Only Medicine.
- 200 Tablets, hospital use only pack
- 60 tablets pack
- 50 tablets pack
- 30 tablets pack
- 100 Tablets, hospital use only pack
- 90 tablets pack
Over 65 ?
(AHT)
This medicine contains the active ingredients:
If you are over 65 years of age, there may be specific risks and recommendations for use of this medicine. Please discuss your individual circumstances with your pharmacist, doctor or health professional. For more information read our page on medication safety for older people.
Pregnant or planning a pregnancy ?
(AHT)
For the active ingredient diazepam
You should seek advice from your doctor or pharmacist about taking this medicine. They can help you balance the risks and the benefits of this medicine during pregnancy.
Reporting side effects
You can help ensure medicines are safe by reporting the side effects you experience.
You can report side effects to your doctor, or directly at www.tga.gov.au/reporting-problems









