Flecatab TM
Listen to the Pronunciation:
You should seek medical advice in relation to medicines and use only as directed by a healthcare professional. Always read the label. If symptoms persist see your healthcare professional.
Active ingredients: flecainide
Brand name
(ARTG)
: FLECATAB flecainide acetate 100mg tablet blister packConsumer Medicine Information (CMI)
Read the CMI leaflet for facts you need to know before, during and after taking your medicine.
Listen to the audio transcription of the CMI leaflet.
For more information about CMIs and how to read them, please visit How to read Consumer Medicine Information (CMI).
What this medicine is used for
(ARTG)
Supraventricular arrhythmias, due to pre-excitation syndromes (e.g Wolff-Parkinson-White and Lown-Ganong-Levine syndromes) or to dual atrioventricular nodal pathways in patients with disabling symptoms; paroxysmal atrial flutter/fibrillation (PAF) associated with disabling symptoms. Although flecainide acetate may be effective in supraventricular arrhythmias in patients with structural heart disease, its use has been associated with life-threatening and occasionally fatal ventricular arrhythmias. In these patients, particularly in the presence of impaired left ventricular function, Flecatab should be used with extreme caution, preferably after other antiarrhythmic drugs have been tried or considered inappropriate. Use of flecainide acetate in chronic atrial fibrillation has not been adequately studied and is not recommended. Life-threatening ventricular arrhythmias not controlled by other drugs. An intravenous form of flecainide acetate is indicated when rapid control or short-term prophylaxis of the above arrhythmias is the main clinical requirement. All use of the injection (available in other brands) should be in hospitals only. Flecatab is used for continuous maintenance of normal rhythm following initial oral or intravenous therapy or conversion by other means. See Warnings and Precautions.
How to use this medicine
(ARTG)
This medicine contains one component only.
Component :
- Tablet, uncoated
- Oral
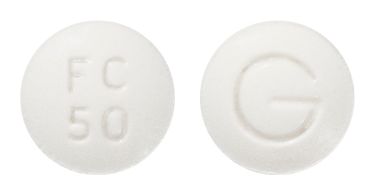
- 8.5mm normal convex white tablet, debossed FC/100 on one side, G on the reverse.
Images
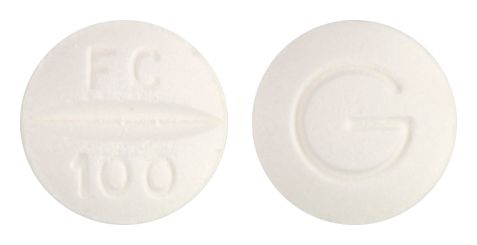

Images © Medicines Information Pty Ltd.
Storage conditions
(ARTG)
- Store below 30 degrees Celsius
- Protect from Light
- Shelf lifetime is 3 Years.
Do I need a prescription ?
(ARTG)
These medicine packs are available from a pharmacist and requires a prescription. It is
- 60 tablets pack
Is this medicine subsidised ?
(PBS)
This medicine was verified as being available on the PBS (Pharmaceutical Benefits Scheme) on May, 1 2026. To learn more about this subsidy, visit the Pharmaceutical Benefits Scheme (PBS) website.
Pregnant or planning a pregnancy ?
(AHT)
For the active ingredient flecainide
You should seek advice from your doctor or pharmacist about taking this medicine. They can help you balance the risks and the benefits of this medicine during pregnancy.
Reporting side effects
You can help ensure medicines are safe by reporting the side effects you experience.
You can report side effects to your doctor, or directly at www.tga.gov.au/reporting-problems