Femoston 2/10 TM
Listen to the Pronunciation:
You should seek medical advice in relation to medicines and use only as directed by a healthcare professional. Always read the label. If symptoms persist see your healthcare professional.
Active ingredients: estradiol + dydrogesterone
Pack: Femoston 2/10 (14 x 2 mg tablets, 14 x 2 mg/10 mg tablets), 28, blister pack
Brand name
(ARTG)
: FEMOSTON 2/10 estradiol 2mg and estradiol 2mg with dydrogesterone 10mg tablet blister pack composite packConsumer Medicine Information (CMI)
Read the CMI leaflet for facts you need to know before, during and after taking your medicine.
For more information about CMIs and how to read them, please visit How to read Consumer Medicine Information (CMI).
What this medicine is used for
(ARTG)
Hormone Replacement therapy (HRT) in estrogen deficiency associated with natural or artificial menopause in women with an intact uterus. Prevention of postmenopausal bone mineral density loss in women. For initiation and continuation of treatment of postmenopausal symptoms, the lowest effective dose for the shortest duration should be used with the goal being short term use (See DOSAGE AND ADMINISTRATION and CLINICAL TRIALS). When prescribed solely for the prevention of postmenopausal bone mineral density loss in women, therapy should only be prescribed for women who are at high risk of future fracture and who are intolerant of, or contraindicated for non-estrogen products approved for prevention of osteoporosis. Life style modifications and the risk benefit profile of FEMOSTON should be taken into careful consideration and discussed with the patient, to allow the patient to make an informed decision prior to prescribing( See PRECAUTIONS and DOSAGE AND ADMINISTRATION).
How to use this medicine
(ARTG)
This medicine is a pack that contains more than one component.
Component :
- Tablet, film coated
- Oral
- Round, biconvex, brick-red film-coated tablet, bearing the inscription 379 on one side.
Component :
- Tablet, film coated
- Oral
- Round, biconvex, yellow, film-coated tablet, bearing the inscription 379 on one side.
Images


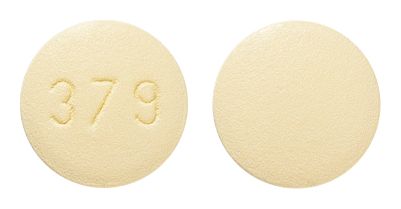

Images © Medicines Information Pty Ltd.
Storage conditions
(ARTG)
- Store below 30 degrees Celsius
- Shelf lifetime is 3 Years.
Do I need a prescription ?
(ARTG)
These medicine packs are available from a pharmacist and requires a prescription. It is
- 28 tablets (14 of each tablet) pack
- 84 tablets (42 of each tablet) pack
Is this medicine subsidised ?
(PBS)
This medicine was verified as being available on the PBS (Pharmaceutical Benefits Scheme) on May, 1 2026. To learn more about this subsidy, visit the Pharmaceutical Benefits Scheme (PBS) website.
Over 65 ?
(AHT)
This medicine contains the active ingredients:
If you are over 65 years of age, there may be specific risks and recommendations for use of this medicine. Please discuss your individual circumstances with your pharmacist, doctor or health professional. For more information read our page on medication safety for older people.
Pregnant or planning a pregnancy ?
(AHT)
For the active ingredient estradiol + dydrogesterone
You should seek advice from your doctor or pharmacist about taking this medicine. They can help you balance the risks and the benefits of this medicine during pregnancy.
Reporting side effects
You can help ensure medicines are safe by reporting the side effects you experience.
You can report side effects to your doctor, or directly at www.tga.gov.au/reporting-problems









