Dilantin TM
Listen to the Pronunciation:
You should seek medical advice in relation to medicines and use only as directed by a healthcare professional. Always read the label. If symptoms persist see your healthcare professional.
Active ingredients: phenytoin
Brand name
(ARTG)
: DILANTIN phenytoin sodium 100 mg capsule bottleConsumer Medicine Information (CMI)
Read the CMI leaflet for facts you need to know before, during and after taking your medicine.
Listen to the audio transcription of the CMI leaflet.
For more information about CMIs and how to read them, please visit How to read Consumer Medicine Information (CMI).
What this medicine is used for
(ARTG)
DILANTIN is indicated for the control of grand mal and psychomotor seizures. DILANTIN will prevent or effectively decrease the incidence and severity of convulsive seizures in a high percentage of cases, with patients exhibiting little tendency to become resistant to its action. Besides its effectiveness in controlling seizures, DILANTIN frequently improves the mental condition and outlook of epileptic patients and there is also increasing evidence that DILANTIN is valuable in the prevention of seizures occurring during or after neurosurgery. Phenytoin serum level determinations may be necessary for optimal dosage adjustments (see Dosage and Administration).
How to use this medicine
(ARTG)
This medicine contains one component only.
Component :
- Capsule, hard
- Oral
- size 3 hard gelatin capsule with white, opaque body and orange cap, printed with a "VTRS" on one side and "VLE 100" on the other in black ink.
Images
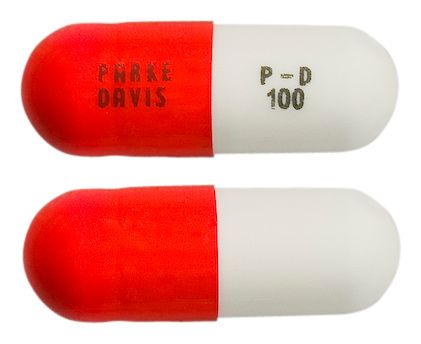
Images © Medicines Information Pty Ltd.
Storage conditions
(ARTG)
- Store below 30 degrees Celsius
- Keep Container Tightly Closed/Airtight
- Shelf lifetime is 24 Months.
Do I need a prescription ?
(ARTG)
These medicine packs are available from a pharmacist and requires a prescription. It is
- 200 pack
Is this medicine subsidised ?
(PBS)
This medicine was verified as being available on the PBS (Pharmaceutical Benefits Scheme) on May, 1 2026. To learn more about this subsidy, visit the Pharmaceutical Benefits Scheme (PBS) website.
Pregnant or planning a pregnancy ?
(AHT)
For the active ingredient phenytoin
You should seek advice from your doctor or pharmacist about taking this medicine. They can help you balance the risks and the benefits of this medicine during pregnancy.
Reporting side effects
You can help ensure medicines are safe by reporting the side effects you experience.
You can report side effects to your doctor, or directly at www.tga.gov.au/reporting-problems










