Ritalin LA TM
Listen to the Pronunciation:
You should seek medical advice in relation to medicines and use only as directed by a healthcare professional. Always read the label. If symptoms persist see your healthcare professional.
Active ingredients: methylphenidate
Brand name
(ARTG)
: RITALIN LA methylphenidate hydrochloride 60mg modified release capsules bottleConsumer Medicine Information (CMI)
Read the CMI leaflet for facts you need to know before, during and after taking your medicine.
For more information about CMIs and how to read them, please visit How to read Consumer Medicine Information (CMI).
What this medicine is used for
(ARTG)
Ritalin 10 tablets and Ritalin LA capsules are indicated for the treatment of ADHD. Ritalin 10 tablets are also indicated for the treatment of narcolepsy. Attention-Deficit Hyperactivity Disorder (ADHD) ADHD was previously known as attention-deficit disorder. Other terms used to describe this behavioural syndrome include: minimal brain dysfunction in children, hyperkinetic child syndrome, minimal brain damage, minimal cerebral dysfunction, minor cerebral dysfunction and psycho-organic syndrome of children. Ritalin 10 / Ritalin LA are indicated as an integral part of a total treatment program for ADHD that may include other measures (psychological, educational and social) for patients with this syndrome. Stimulants are not intended for use in the patient who exhibits symptoms secondary to environmental factors and/or other primary psychiatric disorders, including psychosis. Special diagnostic considerations for ADHD in children: The aetiology of this syndrome is unknown and there is no single diagnostic test. Adequate diagnosis requires the use, not only of medical, but also of psychological, educational and social resources. Characteristics commonly reported include: chronic history of short attention span, distractibility, emotional lability, impulsivity, moderate to severe hyperactivity, minor neurological signs and an abnormal EEG. Learning may or may not be impaired. The diagnosis must be based upon a complete history and evaluation of the child and not solely on the presence of one or more of these characteristics. Drug treatment is not indicated for all children with this syndrome. Stimulants are not intended for use in children who exhibit symptoms secondary to environmental factors (e.g. child abuse in particular) or primary psychiatric disorders. Appropriate educational placement is essential and psychosocial intervention is generally necessary. When remedial measures alone are insufficient, the decision to prescribe stimulant medicine will depend upon the physician's assessment of the chronicity and severity of the child's symptoms. Continuation of treatment in adolescent and special diagnostic considerations for ADHD in adults: There is limited information to guide clinicians about how long older adolescents should continue to receive treatment with drugs for attention deficit hyperactivity disorder (ADHD). The decision should be based on the extent to which symptoms of ADHD and social functioning have improved to a point that medication is no longer needed. If older adolescents have been largely symptom-free for a year and are functioning well, a trial without medication is warranted. This should be undertaken at times of low stress such as during holidays or in a period when a school routine is well established. ADHD needs to be considered in adults who present with longstanding symptoms suggestive of ADHD (inattention, impulsivity, disorganisation) that appear to have started in childhood and are persisting into adult life. Further, people with personality disorder and/or problems with drug use accompanied by a significant level of impulsivity and inattention should be referred for evaluation by a psychiatrist with the training and skills required to assess and treat ADHD. This expertise is necessary due to the overlap of ADHD symptoms with anxiety, mood and personality disorders. Narcolepsy The symptoms include daytime sleepiness, inappropriate sleep episodes and rapidly occurring loss of voluntary muscle tone. Ritalin 10 is effective for symptoms of sleepiness but not for loss of voluntary muscle tone.
How to use this medicine
(ARTG)
This medicine contains one component only.
Component :
- Capsule, modified release
- Oral
- Size 00, light brown opaque gelatin cap and a yellow opaque body, imprinted with NVR on the cap and R60 on the body in tan-coloured ink, containing white to off-white beads that are roughly spherical in shape
Storage conditions
(ARTG)
- Store below 30 degrees Celsius
- Keep Container Tightly Closed/Airtight
- Shelf lifetime is 18 Months.
Do I need a prescription ?
(ARTG)
These medicines require authorisation for prescription from your doctor. It is Schedule 8 : Controlled Drug.
- 100 capsules pack
- 30 capsules pack
Is this medicine subsidised ?
(PBS)
This medicine was verified as being available on the PBS (Pharmaceutical Benefits Scheme) on May, 1 2026. To learn more about this subsidy, visit the Pharmaceutical Benefits Scheme (PBS) website.
Pregnant or planning a pregnancy ?
(AHT)
For the active ingredient methylphenidate
You should seek advice from your doctor or pharmacist about taking this medicine. They can help you balance the risks and the benefits of this medicine during pregnancy.
Reporting side effects
You can help ensure medicines are safe by reporting the side effects you experience.
You can report side effects to your doctor, or directly at www.tga.gov.au/reporting-problems
Brand name
(ARTG)
: RITALIN LA methylphenidate hydrochloride 60mg modified release capsules bottleConsumer Medicine Information (CMI)
Read the CMI leaflet for facts you need to know before, during and after taking your medicine.
For more information about CMIs and how to read them, please visit How to read Consumer Medicine Information (CMI).
What this medicine is used for
(ARTG)
Ritalin 10 tablets and Ritalin LA capsules are indicated for the treatment of ADHD. Ritalin 10 tablets are also indicated for the treatment of narcolepsy. Attention-Deficit Hyperactivity Disorder (ADHD) ADHD was previously known as attention-deficit disorder. Other terms used to describe this behavioural syndrome include: minimal brain dysfunction in children, hyperkinetic child syndrome, minimal brain damage, minimal cerebral dysfunction, minor cerebral dysfunction and psycho-organic syndrome of children. Ritalin 10 / Ritalin LA are indicated as an integral part of a total treatment program for ADHD that may include other measures (psychological, educational and social) for patients with this syndrome. Stimulants are not intended for use in the patient who exhibits symptoms secondary to environmental factors and/or other primary psychiatric disorders, including psychosis. Special diagnostic considerations for ADHD in children: The aetiology of this syndrome is unknown and there is no single diagnostic test. Adequate diagnosis requires the use, not only of medical, but also of psychological, educational and social resources. Characteristics commonly reported include: chronic history of short attention span, distractibility, emotional lability, impulsivity, moderate to severe hyperactivity, minor neurological signs and an abnormal EEG. Learning may or may not be impaired. The diagnosis must be based upon a complete history and evaluation of the child and not solely on the presence of one or more of these characteristics. Drug treatment is not indicated for all children with this syndrome. Stimulants are not intended for use in children who exhibit symptoms secondary to environmental factors (e.g. child abuse in particular) or primary psychiatric disorders. Appropriate educational placement is essential and psychosocial intervention is generally necessary. When remedial measures alone are insufficient, the decision to prescribe stimulant medicine will depend upon the physician's assessment of the chronicity and severity of the child's symptoms. Continuation of treatment in adolescent and special diagnostic considerations for ADHD in adults: There is limited information to guide clinicians about how long older adolescents should continue to receive treatment with drugs for attention deficit hyperactivity disorder (ADHD). The decision should be based on the extent to which symptoms of ADHD and social functioning have improved to a point that medication is no longer needed. If older adolescents have been largely symptom-free for a year and are functioning well, a trial without medication is warranted. This should be undertaken at times of low stress such as during holidays or in a period when a school routine is well established. ADHD needs to be considered in adults who present with longstanding symptoms suggestive of ADHD (inattention, impulsivity, disorganisation) that appear to have started in childhood and are persisting into adult life. Further, people with personality disorder and/or problems with drug use accompanied by a significant level of impulsivity and inattention should be referred for evaluation by a psychiatrist with the training and skills required to assess and treat ADHD. This expertise is necessary due to the overlap of ADHD symptoms with anxiety, mood and personality disorders. Narcolepsy The symptoms include daytime sleepiness, inappropriate sleep episodes and rapidly occurring loss of voluntary muscle tone. Ritalin 10 is effective for symptoms of sleepiness but not for loss of voluntary muscle tone.
How to use this medicine
(ARTG)
This medicine contains one component only.
Component :
- Capsule, modified release
- Oral
- Size 00, light brown opaque gelatin cap and a yellow opaque body, imprinted with NVR on the cap and R60 on the body in tan-coloured ink, containing white to off-white beads that are roughly spherical in shape
Storage conditions
(ARTG)
- Store below 30 degrees Celsius
- Keep Container Tightly Closed/Airtight
- Shelf lifetime is 18 Months.
Do I need a prescription ?
(ARTG)
These medicines require authorisation for prescription from your doctor. It is Schedule 8 : Controlled Drug.
- 100 capsules pack
- 30 capsules pack
Pregnant or planning a pregnancy ?
(AHT)
For the active ingredient methylphenidate
You should seek advice from your doctor or pharmacist about taking this medicine. They can help you balance the risks and the benefits of this medicine during pregnancy.
Reporting side effects
You can help ensure medicines are safe by reporting the side effects you experience.
You can report side effects to your doctor, or directly at www.tga.gov.au/reporting-problems
Brand name
(ARTG)
: RITALIN LA methylphenidate hydrochloride 20mg modified release capsule bottleConsumer Medicine Information (CMI)
Read the CMI leaflet for facts you need to know before, during and after taking your medicine.
For more information about CMIs and how to read them, please visit How to read Consumer Medicine Information (CMI).
What this medicine is used for
(ARTG)
Ritalin 10 tablets and Ritalin LA capsules are indicated for the treatment of ADHD. Ritalin 10 tablets are also indicated for the treatment of narcolepsy. Attention-Deficit Hyperactivity Disorder (ADHD) ADHD was previously known as attention-deficit disorder. Other terms used to describe this behavioural syndrome include: minimal brain dysfunction in children, hyperkinetic child syndrome, minimal brain damage, minimal cerebral dysfunction, minor cerebral dysfunction and psycho-organic syndrome of children. Ritalin 10 / Ritalin LA are indicatedas an integral part of a total treatment program for ADHD that may include other measures (psychological, educational and social) for patients with this syndrome. Stimulants are not intended for use in the patient who exhibits symptoms secondary to environmental factors and/or other primary psychiatric disorders, including psychosis. Special diagnostic considerations for ADHD in children: The aetiology of this syndrome is unknown and there is no single diagnostic test. Adequate diagnosis requires the use, not only of medical, but also of psychological, educational and social resources. Characteristics commonly reported include: chronic history of short attention span, distractibility, emotional lability, impulsivity, moderate to severe hyperactivity, minor neurological signs and an abnormal EEG. Learning may or may not be impaired. The diagnosis must be based upon a complete history and evaluation of the child and not solely on the presence of one or more of these characteristics. Drug treatment is not indicated for all children with this syndrome. Stimulants are not intended for use in children who exhibit symptoms secondary to environmental factors (e.g. child abuse in particular) or primary psychiatric disorders. Appropriate educational placement is essential and psychosocial intervention is generally necessary. When remedial measures alone are insufficient, the decision to prescribe stimulant medicine will depend upon the physician's assessment of the chronicity and severity of the child's symptoms. Continuation of treatment in adolescent and special diagnostic considerations for ADHD in adults: There is limited information to guide clinicians about how long older adolescents should continue to receive treatment with drugs for attention deficit hyperactivity disorder (ADHD). The decision should be based on the extent to which symptoms of ADHD and social functioning have improved to a point that medication is no longer needed. If older adolescents have been largely symptom-free for a year and are functioning well, a trial without medication is warranted. This should be undertaken at times of low stress such as during holidays or in a period when a school routine is well established. ADHD needs to be considered in adults who present with longstanding symptoms suggestive of ADHD (inattention, impulsivity, disorganisation) that appear to have started in childhood and are persisting into adult life. Further, people with personality disorder and/or problems with drug use accompanied by a significant level of impulsivity and inattention should be referred for evaluation by a psychiatrist with the training and skills required to assess and treat ADHD. This expertise is necessary due to the overlap of ADHD symptoms with anxiety, mood and personality disorders. Narcolepsy The symptoms include daytime sleepiness, inappropriate sleep episodes and rapidly occurring loss of voluntary muscle tone. Ritalin 10 is effective for symptoms of sleepiness but not for loss of voluntary muscle tone.
How to use this medicine
(ARTG)
This medicine contains one component only.
Component :
- Capsule, modified release
- Oral
- WHITE TO OFF-WHITE BEADS IN A WHITE OPAQUE HARD GELATIN CAPSULE WITH "NVR" ON CAP AND "R20" ON THE BODY IN TAN INK
Images
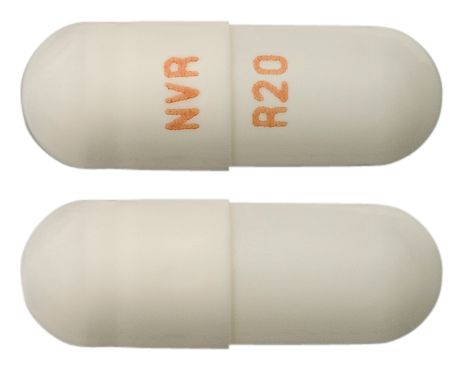
Images © Medicines Information Pty Ltd.
Storage conditions
(ARTG)
- Store below 30 degrees Celsius
- Keep Container Tightly Closed/Airtight
- Shelf lifetime is 24 Months.
Do I need a prescription ?
(ARTG)
These medicines require authorisation for prescription from your doctor. It is Schedule 8 : Controlled Drug.
- 100 capsules pack
- 30 capsules pack
Pregnant or planning a pregnancy ?
(AHT)
For the active ingredient methylphenidate
You should seek advice from your doctor or pharmacist about taking this medicine. They can help you balance the risks and the benefits of this medicine during pregnancy.
Reporting side effects
You can help ensure medicines are safe by reporting the side effects you experience.
You can report side effects to your doctor, or directly at www.tga.gov.au/reporting-problems
Brand name
(ARTG)
: RITALIN LA methylphenidate hydrochloride 20mg modified release capsule bottleConsumer Medicine Information (CMI)
Read the CMI leaflet for facts you need to know before, during and after taking your medicine.
For more information about CMIs and how to read them, please visit How to read Consumer Medicine Information (CMI).
What this medicine is used for
(ARTG)
Ritalin 10 tablets and Ritalin LA capsules are indicated for the treatment of ADHD. Ritalin 10 tablets are also indicated for the treatment of narcolepsy. Attention-Deficit Hyperactivity Disorder (ADHD) ADHD was previously known as attention-deficit disorder. Other terms used to describe this behavioural syndrome include: minimal brain dysfunction in children, hyperkinetic child syndrome, minimal brain damage, minimal cerebral dysfunction, minor cerebral dysfunction and psycho-organic syndrome of children. Ritalin 10 / Ritalin LA are indicatedas an integral part of a total treatment program for ADHD that may include other measures (psychological, educational and social) for patients with this syndrome. Stimulants are not intended for use in the patient who exhibits symptoms secondary to environmental factors and/or other primary psychiatric disorders, including psychosis. Special diagnostic considerations for ADHD in children: The aetiology of this syndrome is unknown and there is no single diagnostic test. Adequate diagnosis requires the use, not only of medical, but also of psychological, educational and social resources. Characteristics commonly reported include: chronic history of short attention span, distractibility, emotional lability, impulsivity, moderate to severe hyperactivity, minor neurological signs and an abnormal EEG. Learning may or may not be impaired. The diagnosis must be based upon a complete history and evaluation of the child and not solely on the presence of one or more of these characteristics. Drug treatment is not indicated for all children with this syndrome. Stimulants are not intended for use in children who exhibit symptoms secondary to environmental factors (e.g. child abuse in particular) or primary psychiatric disorders. Appropriate educational placement is essential and psychosocial intervention is generally necessary. When remedial measures alone are insufficient, the decision to prescribe stimulant medicine will depend upon the physician's assessment of the chronicity and severity of the child's symptoms. Continuation of treatment in adolescent and special diagnostic considerations for ADHD in adults: There is limited information to guide clinicians about how long older adolescents should continue to receive treatment with drugs for attention deficit hyperactivity disorder (ADHD). The decision should be based on the extent to which symptoms of ADHD and social functioning have improved to a point that medication is no longer needed. If older adolescents have been largely symptom-free for a year and are functioning well, a trial without medication is warranted. This should be undertaken at times of low stress such as during holidays or in a period when a school routine is well established. ADHD needs to be considered in adults who present with longstanding symptoms suggestive of ADHD (inattention, impulsivity, disorganisation) that appear to have started in childhood and are persisting into adult life. Further, people with personality disorder and/or problems with drug use accompanied by a significant level of impulsivity and inattention should be referred for evaluation by a psychiatrist with the training and skills required to assess and treat ADHD. This expertise is necessary due to the overlap of ADHD symptoms with anxiety, mood and personality disorders. Narcolepsy The symptoms include daytime sleepiness, inappropriate sleep episodes and rapidly occurring loss of voluntary muscle tone. Ritalin 10 is effective for symptoms of sleepiness but not for loss of voluntary muscle tone.
How to use this medicine
(ARTG)
This medicine contains one component only.
Component :
- Capsule, modified release
- Oral
- WHITE TO OFF-WHITE BEADS IN A WHITE OPAQUE HARD GELATIN CAPSULE WITH "NVR" ON CAP AND "R20" ON THE BODY IN TAN INK
Images

Images © Medicines Information Pty Ltd.
Storage conditions
(ARTG)
- Store below 30 degrees Celsius
- Keep Container Tightly Closed/Airtight
- Shelf lifetime is 24 Months.
Do I need a prescription ?
(ARTG)
These medicines require authorisation for prescription from your doctor. It is Schedule 8 : Controlled Drug.
- 100 capsules pack
- 30 capsules pack
Is this medicine subsidised ?
(PBS)
This medicine was verified as being available on the PBS (Pharmaceutical Benefits Scheme) on May, 1 2026. To learn more about this subsidy, visit the Pharmaceutical Benefits Scheme (PBS) website.
Pregnant or planning a pregnancy ?
(AHT)
For the active ingredient methylphenidate
You should seek advice from your doctor or pharmacist about taking this medicine. They can help you balance the risks and the benefits of this medicine during pregnancy.
Reporting side effects
You can help ensure medicines are safe by reporting the side effects you experience.
You can report side effects to your doctor, or directly at www.tga.gov.au/reporting-problems
Brand name
(ARTG)
: RITALIN LA methylphenidate hydrochloride 30mg modified release capsule bottleConsumer Medicine Information (CMI)
Read the CMI leaflet for facts you need to know before, during and after taking your medicine.
For more information about CMIs and how to read them, please visit How to read Consumer Medicine Information (CMI).
What this medicine is used for
(ARTG)
Ritalin 10 tablets and Ritalin LA capsules are indicated for the treatment of ADHD. Ritalin 10 tablets are also indicated for the treatment of narcolepsy. Attention-Deficit Hyperactivity Disorder (ADHD) ADHD was previously known as attention-deficit disorder. Other terms used to describe this behavioural syndrome include: minimal brain dysfunction in children, hyperkinetic child syndrome, minimal brain damage, minimal cerebral dysfunction, minor cerebral dysfunction and psycho-organic syndrome of children. Ritalin 10 / Ritalin LA are indicatedas an integral part of a total treatment program for ADHD that may include other measures (psychological, educational and social) for patients with this syndrome. Stimulants are not intended for use in the patient who exhibits symptoms secondary to environmental factors and/or other primary psychiatric disorders, including psychosis. Special diagnostic considerations for ADHD in children: The aetiology of this syndrome is unknown and there is no single diagnostic test. Adequate diagnosis requires the use, not only of medical, but also of psychological, educational and social resources. Characteristics commonly reported include: chronic history of short attention span, distractibility, emotional lability, impulsivity, moderate to severe hyperactivity, minor neurological signs and an abnormal EEG. Learning may or may not be impaired. The diagnosis must be based upon a complete history and evaluation of the child and not solely on the presence of one or more of these characteristics. Drug treatment is not indicated for all children with this syndrome. Stimulants are not intended for use in children who exhibit symptoms secondary to environmental factors (e.g. child abuse in particular) or primary psychiatric disorders. Appropriate educational placement is essential and psychosocial intervention is generally necessary. When remedial measures alone are insufficient, the decision to prescribe stimulant medicine will depend upon the physician's assessment of the chronicity and severity of the child's symptoms. Continuation of treatment in adolescent and special diagnostic considerations for ADHD in adults: There is limited information to guide clinicians about how long older adolescents should continue to receive treatment with drugs for attention deficit hyperactivity disorder (ADHD). The decision should be based on the extent to which symptoms of ADHD and social functioning have improved to a point that medication is no longer needed. If older adolescents have been largely symptom-free for a year and are functioning well, a trial without medication is warranted. This should be undertaken at times of low stress such as during holidays or in a period when a school routine is well established. ADHD needs to be considered in adults who present with longstanding symptoms suggestive of ADHD (inattention, impulsivity, disorganisation) that appear to have started in childhood and are persisting into adult life. Further, people with personality disorder and/or problems with drug use accompanied by a significant level of impulsivity and inattention should be referred for evaluation by a psychiatrist with the training and skills required to assess and treat ADHD. This expertise is necessary due to the overlap of ADHD symptoms with anxiety, mood and personality disorders. Narcolepsy The symptoms include daytime sleepiness, inappropriate sleep episodes and rapidly occurring loss of voluntary muscle tone. Ritalin 10 is effective for symptoms of sleepiness but not for loss of voluntary muscle tone.
How to use this medicine
(ARTG)
This medicine contains one component only.
Component :
- Capsule, modified release
- Oral
- WHITE TO OFF-WHITE BEADS IN A YELLOW OPAQUE HARD GELATIN CAPSULE WITH "NVR" ON CAP AND "R30" ON THE BODY IN TAN INK
Images
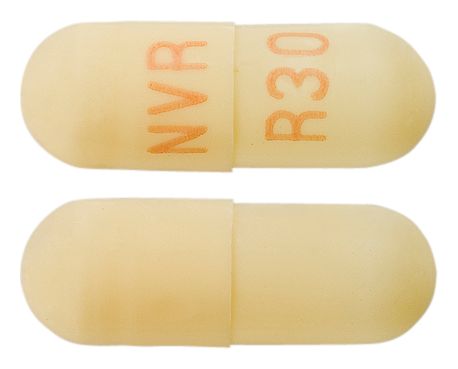
Images © Medicines Information Pty Ltd.
Storage conditions
(ARTG)
- Store below 30 degrees Celsius
- Keep Container Tightly Closed/Airtight
- Shelf lifetime is 24 Months.
Do I need a prescription ?
(ARTG)
These medicines require authorisation for prescription from your doctor. It is Schedule 8 : Controlled Drug.
- 100 capsules pack
- 30 capsules pack
Pregnant or planning a pregnancy ?
(AHT)
For the active ingredient methylphenidate
You should seek advice from your doctor or pharmacist about taking this medicine. They can help you balance the risks and the benefits of this medicine during pregnancy.
Reporting side effects
You can help ensure medicines are safe by reporting the side effects you experience.
You can report side effects to your doctor, or directly at www.tga.gov.au/reporting-problems
Brand name
(ARTG)
: RITALIN LA methylphenidate hydrochloride 30mg modified release capsule bottleConsumer Medicine Information (CMI)
Read the CMI leaflet for facts you need to know before, during and after taking your medicine.
For more information about CMIs and how to read them, please visit How to read Consumer Medicine Information (CMI).
What this medicine is used for
(ARTG)
Ritalin 10 tablets and Ritalin LA capsules are indicated for the treatment of ADHD. Ritalin 10 tablets are also indicated for the treatment of narcolepsy. Attention-Deficit Hyperactivity Disorder (ADHD) ADHD was previously known as attention-deficit disorder. Other terms used to describe this behavioural syndrome include: minimal brain dysfunction in children, hyperkinetic child syndrome, minimal brain damage, minimal cerebral dysfunction, minor cerebral dysfunction and psycho-organic syndrome of children. Ritalin 10 / Ritalin LA are indicatedas an integral part of a total treatment program for ADHD that may include other measures (psychological, educational and social) for patients with this syndrome. Stimulants are not intended for use in the patient who exhibits symptoms secondary to environmental factors and/or other primary psychiatric disorders, including psychosis. Special diagnostic considerations for ADHD in children: The aetiology of this syndrome is unknown and there is no single diagnostic test. Adequate diagnosis requires the use, not only of medical, but also of psychological, educational and social resources. Characteristics commonly reported include: chronic history of short attention span, distractibility, emotional lability, impulsivity, moderate to severe hyperactivity, minor neurological signs and an abnormal EEG. Learning may or may not be impaired. The diagnosis must be based upon a complete history and evaluation of the child and not solely on the presence of one or more of these characteristics. Drug treatment is not indicated for all children with this syndrome. Stimulants are not intended for use in children who exhibit symptoms secondary to environmental factors (e.g. child abuse in particular) or primary psychiatric disorders. Appropriate educational placement is essential and psychosocial intervention is generally necessary. When remedial measures alone are insufficient, the decision to prescribe stimulant medicine will depend upon the physician's assessment of the chronicity and severity of the child's symptoms. Continuation of treatment in adolescent and special diagnostic considerations for ADHD in adults: There is limited information to guide clinicians about how long older adolescents should continue to receive treatment with drugs for attention deficit hyperactivity disorder (ADHD). The decision should be based on the extent to which symptoms of ADHD and social functioning have improved to a point that medication is no longer needed. If older adolescents have been largely symptom-free for a year and are functioning well, a trial without medication is warranted. This should be undertaken at times of low stress such as during holidays or in a period when a school routine is well established. ADHD needs to be considered in adults who present with longstanding symptoms suggestive of ADHD (inattention, impulsivity, disorganisation) that appear to have started in childhood and are persisting into adult life. Further, people with personality disorder and/or problems with drug use accompanied by a significant level of impulsivity and inattention should be referred for evaluation by a psychiatrist with the training and skills required to assess and treat ADHD. This expertise is necessary due to the overlap of ADHD symptoms with anxiety, mood and personality disorders. Narcolepsy The symptoms include daytime sleepiness, inappropriate sleep episodes and rapidly occurring loss of voluntary muscle tone. Ritalin 10 is effective for symptoms of sleepiness but not for loss of voluntary muscle tone.
How to use this medicine
(ARTG)
This medicine contains one component only.
Component :
- Capsule, modified release
- Oral
- WHITE TO OFF-WHITE BEADS IN A YELLOW OPAQUE HARD GELATIN CAPSULE WITH "NVR" ON CAP AND "R30" ON THE BODY IN TAN INK
Images

Images © Medicines Information Pty Ltd.
Storage conditions
(ARTG)
- Store below 30 degrees Celsius
- Keep Container Tightly Closed/Airtight
- Shelf lifetime is 24 Months.
Do I need a prescription ?
(ARTG)
These medicines require authorisation for prescription from your doctor. It is Schedule 8 : Controlled Drug.
- 100 capsules pack
- 30 capsules pack
Is this medicine subsidised ?
(PBS)
This medicine was verified as being available on the PBS (Pharmaceutical Benefits Scheme) on May, 1 2026. To learn more about this subsidy, visit the Pharmaceutical Benefits Scheme (PBS) website.
Pregnant or planning a pregnancy ?
(AHT)
For the active ingredient methylphenidate
You should seek advice from your doctor or pharmacist about taking this medicine. They can help you balance the risks and the benefits of this medicine during pregnancy.
Reporting side effects
You can help ensure medicines are safe by reporting the side effects you experience.
You can report side effects to your doctor, or directly at www.tga.gov.au/reporting-problems
Brand name
(ARTG)
: RITALIN LA methylphenidate hydrochloride 40mg modified release capsule bottleConsumer Medicine Information (CMI)
Read the CMI leaflet for facts you need to know before, during and after taking your medicine.
For more information about CMIs and how to read them, please visit How to read Consumer Medicine Information (CMI).
What this medicine is used for
(ARTG)
Ritalin 10 tablets and Ritalin LA capsules are indicated for the treatment of ADHD. Ritalin 10 tablets are also indicated for the treatment of narcolepsy. Attention-Deficit Hyperactivity Disorder (ADHD) ADHD was previously known as attention-deficit disorder. Other terms used to describe this behavioural syndrome include: minimal brain dysfunction in children, hyperkinetic child syndrome, minimal brain damage, minimal cerebral dysfunction, minor cerebral dysfunction and psycho-organic syndrome of children. Ritalin 10 / Ritalin LA are indicatedas an integral part of a total treatment program for ADHD that may include other measures (psychological, educational and social) for patients with this syndrome. Stimulants are not intended for use in the patient who exhibits symptoms secondary to environmental factors and/or other primary psychiatric disorders, including psychosis. Special diagnostic considerations for ADHD in children: The aetiology of this syndrome is unknown and there is no single diagnostic test. Adequate diagnosis requires the use, not only of medical, but also of psychological, educational and social resources. Characteristics commonly reported include: chronic history of short attention span, distractibility, emotional lability, impulsivity, moderate to severe hyperactivity, minor neurological signs and an abnormal EEG. Learning may or may not be impaired. The diagnosis must be based upon a complete history and evaluation of the child and not solely on the presence of one or more of these characteristics. Drug treatment is not indicated for all children with this syndrome. Stimulants are not intended for use in children who exhibit symptoms secondary to environmental factors (e.g. child abuse in particular) or primary psychiatric disorders. Appropriate educational placement is essential and psychosocial intervention is generally necessary. When remedial measures alone are insufficient, the decision to prescribe stimulant medicine will depend upon the physician's assessment of the chronicity and severity of the child's symptoms. Continuation of treatment in adolescent and special diagnostic considerations for ADHD in adults: There is limited information to guide clinicians about how long older adolescents should continue to receive treatment with drugs for attention deficit hyperactivity disorder (ADHD). The decision should be based on the extent to which symptoms of ADHD and social functioning have improved to a point that medication is no longer needed. If older adolescents have been largely symptom-free for a year and are functioning well, a trial without medication is warranted. This should be undertaken at times of low stress such as during holidays or in a period when a school routine is well established. ADHD needs to be considered in adults who present with longstanding symptoms suggestive of ADHD (inattention, impulsivity, disorganisation) that appear to have started in childhood and are persisting into adult life. Further, people with personality disorder and/or problems with drug use accompanied by a significant level of impulsivity and inattention should be referred for evaluation by a psychiatrist with the training and skills required to assess and treat ADHD. This expertise is necessary due to the overlap of ADHD symptoms with anxiety, mood and personality disorders. Narcolepsy The symptoms include daytime sleepiness, inappropriate sleep episodes and rapidly occurring loss of voluntary muscle tone. Ritalin 10 is effective for symptoms of sleepiness but not for loss of voluntary muscle tone.
How to use this medicine
(ARTG)
This medicine contains one component only.
Component :
- Capsule, modified release
- Oral
- WHITE TO OFF-WHITE BEADS IN A LIGHT BROWN OPAQUE HARD GELATIN CAPSULE WITH "NVR" ON CAP AND "R40" ON THE BODY IN TAN INK
Images

Images © Medicines Information Pty Ltd.
Storage conditions
(ARTG)
- Store below 30 degrees Celsius
- Keep Container Tightly Closed/Airtight
- Shelf lifetime is 24 Months.
Do I need a prescription ?
(ARTG)
These medicines require authorisation for prescription from your doctor. It is Schedule 8 : Controlled Drug.
- 100 capsules pack
- 30 capsules pack
Pregnant or planning a pregnancy ?
(AHT)
For the active ingredient methylphenidate
You should seek advice from your doctor or pharmacist about taking this medicine. They can help you balance the risks and the benefits of this medicine during pregnancy.
Reporting side effects
You can help ensure medicines are safe by reporting the side effects you experience.
You can report side effects to your doctor, or directly at www.tga.gov.au/reporting-problems
Brand name
(ARTG)
: RITALIN LA methylphenidate hydrochloride 40mg modified release capsule bottleConsumer Medicine Information (CMI)
Read the CMI leaflet for facts you need to know before, during and after taking your medicine.
For more information about CMIs and how to read them, please visit How to read Consumer Medicine Information (CMI).
What this medicine is used for
(ARTG)
Ritalin 10 tablets and Ritalin LA capsules are indicated for the treatment of ADHD. Ritalin 10 tablets are also indicated for the treatment of narcolepsy. Attention-Deficit Hyperactivity Disorder (ADHD) ADHD was previously known as attention-deficit disorder. Other terms used to describe this behavioural syndrome include: minimal brain dysfunction in children, hyperkinetic child syndrome, minimal brain damage, minimal cerebral dysfunction, minor cerebral dysfunction and psycho-organic syndrome of children. Ritalin 10 / Ritalin LA are indicatedas an integral part of a total treatment program for ADHD that may include other measures (psychological, educational and social) for patients with this syndrome. Stimulants are not intended for use in the patient who exhibits symptoms secondary to environmental factors and/or other primary psychiatric disorders, including psychosis. Special diagnostic considerations for ADHD in children: The aetiology of this syndrome is unknown and there is no single diagnostic test. Adequate diagnosis requires the use, not only of medical, but also of psychological, educational and social resources. Characteristics commonly reported include: chronic history of short attention span, distractibility, emotional lability, impulsivity, moderate to severe hyperactivity, minor neurological signs and an abnormal EEG. Learning may or may not be impaired. The diagnosis must be based upon a complete history and evaluation of the child and not solely on the presence of one or more of these characteristics. Drug treatment is not indicated for all children with this syndrome. Stimulants are not intended for use in children who exhibit symptoms secondary to environmental factors (e.g. child abuse in particular) or primary psychiatric disorders. Appropriate educational placement is essential and psychosocial intervention is generally necessary. When remedial measures alone are insufficient, the decision to prescribe stimulant medicine will depend upon the physician's assessment of the chronicity and severity of the child's symptoms. Continuation of treatment in adolescent and special diagnostic considerations for ADHD in adults: There is limited information to guide clinicians about how long older adolescents should continue to receive treatment with drugs for attention deficit hyperactivity disorder (ADHD). The decision should be based on the extent to which symptoms of ADHD and social functioning have improved to a point that medication is no longer needed. If older adolescents have been largely symptom-free for a year and are functioning well, a trial without medication is warranted. This should be undertaken at times of low stress such as during holidays or in a period when a school routine is well established. ADHD needs to be considered in adults who present with longstanding symptoms suggestive of ADHD (inattention, impulsivity, disorganisation) that appear to have started in childhood and are persisting into adult life. Further, people with personality disorder and/or problems with drug use accompanied by a significant level of impulsivity and inattention should be referred for evaluation by a psychiatrist with the training and skills required to assess and treat ADHD. This expertise is necessary due to the overlap of ADHD symptoms with anxiety, mood and personality disorders. Narcolepsy The symptoms include daytime sleepiness, inappropriate sleep episodes and rapidly occurring loss of voluntary muscle tone. Ritalin 10 is effective for symptoms of sleepiness but not for loss of voluntary muscle tone.
How to use this medicine
(ARTG)
This medicine contains one component only.
Component :
- Capsule, modified release
- Oral
- WHITE TO OFF-WHITE BEADS IN A LIGHT BROWN OPAQUE HARD GELATIN CAPSULE WITH "NVR" ON CAP AND "R40" ON THE BODY IN TAN INK
Images

Images © Medicines Information Pty Ltd.
Storage conditions
(ARTG)
- Store below 30 degrees Celsius
- Keep Container Tightly Closed/Airtight
- Shelf lifetime is 24 Months.
Do I need a prescription ?
(ARTG)
These medicines require authorisation for prescription from your doctor. It is Schedule 8 : Controlled Drug.
- 100 capsules pack
- 30 capsules pack
Is this medicine subsidised ?
(PBS)
This medicine was verified as being available on the PBS (Pharmaceutical Benefits Scheme) on May, 1 2026. To learn more about this subsidy, visit the Pharmaceutical Benefits Scheme (PBS) website.
Pregnant or planning a pregnancy ?
(AHT)
For the active ingredient methylphenidate
You should seek advice from your doctor or pharmacist about taking this medicine. They can help you balance the risks and the benefits of this medicine during pregnancy.
Reporting side effects
You can help ensure medicines are safe by reporting the side effects you experience.
You can report side effects to your doctor, or directly at www.tga.gov.au/reporting-problems
Brand name
(ARTG)
: RITALIN LA methylphenidate hydrochloride 10 mg modified release capsule bottleConsumer Medicine Information (CMI)
Read the CMI leaflet for facts you need to know before, during and after taking your medicine.
For more information about CMIs and how to read them, please visit How to read Consumer Medicine Information (CMI).
What this medicine is used for
(ARTG)
Ritalin 10 tablets and Ritalin LA capsules are indicated for the treatment of ADHD. Ritalin 10 tablets are also indicated for the treatment of narcolepsy. Attention-Deficit Hyperactivity Disorder (ADHD) ADHD was previously known as attention-deficit disorder. Other terms used to describe this behavioural syndrome include: minimal brain dysfunction in children, hyperkinetic child syndrome, minimal brain damage, minimal cerebral dysfunction, minor cerebral dysfunction and psycho-organic syndrome of children. Ritalin 10 / Ritalin LA are indicatedas an integral part of a total treatment program for ADHD that may include other measures (psychological, educational and social) for patients with this syndrome. Stimulants are not intended for use in the patient who exhibits symptoms secondary to environmental factors and/or other primary psychiatric disorders, including psychosis. Special diagnostic considerations for ADHD in children: The aetiology of this syndrome is unknown and there is no single diagnostic test. Adequate diagnosis requires the use, not only of medical, but also of psychological, educational and social resources. Characteristics commonly reported include: chronic history of short attention span, distractibility, emotional lability, impulsivity, moderate to severe hyperactivity, minor neurological signs and an abnormal EEG. Learning may or may not be impaired. The diagnosis must be based upon a complete history and evaluation of the child and not solely on the presence of one or more of these characteristics. Drug treatment is not indicated for all children with this syndrome. Stimulants are not intended for use in children who exhibit symptoms secondary to environmental factors (e.g. child abuse in particular) or primary psychiatric disorders. Appropriate educational placement is essential and psychosocial intervention is generally necessary. When remedial measures alone are insufficient, the decision to prescribe stimulant medicine will depend upon the physician's assessment of the chronicity and severity of the child's symptoms. Continuation of treatment in adolescent and special diagnostic considerations for ADHD in adults: There is limited information to guide clinicians about how long older adolescents should continue to receive treatment with drugs for attention deficit hyperactivity disorder (ADHD). The decision should be based on the extent to which symptoms of ADHD and social functioning have improved to a point that medication is no longer needed. If older adolescents have been largely symptom-free for a year and are functioning well, a trial without medication is warranted. This should be undertaken at times of low stress such as during holidays or in a period when a school routine is well established. ADHD needs to be considered in adults who present with longstanding symptoms suggestive of ADHD (inattention, impulsivity, disorganisation) that appear to have started in childhood and are persisting into adult life. Further, people with personality disorder and/or problems with drug use accompanied by a significant level of impulsivity and inattention should be referred for evaluation by a psychiatrist with the training and skills required to assess and treat ADHD. This expertise is necessary due to the overlap of ADHD symptoms with anxiety, mood and personality disorders. Narcolepsy The symptoms include daytime sleepiness, inappropriate sleep episodes and rapidly occurring loss of voluntary muscle tone. Ritalin 10 is effective for symptoms of sleepiness but not for loss of voluntary muscle tone.
How to use this medicine
(ARTG)
This medicine contains one component only.
Component :
- Capsule, modified release
- Oral
- White to off-white beads in light brown opaque hard gelatin capsule with "NVR" on cap and "R10" on a white opaque body in tan ink.
Images
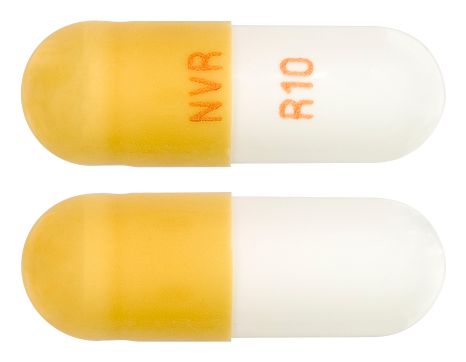
Images © Medicines Information Pty Ltd.
Storage conditions
(ARTG)
- Store below 30 degrees Celsius
- Keep Container Tightly Closed/Airtight
- Shelf lifetime is 24 Months.
Do I need a prescription ?
(ARTG)
These medicines require authorisation for prescription from your doctor. It is Schedule 8 : Controlled Drug.
- 100 pack
- 30 pack
Pregnant or planning a pregnancy ?
(AHT)
For the active ingredient methylphenidate
You should seek advice from your doctor or pharmacist about taking this medicine. They can help you balance the risks and the benefits of this medicine during pregnancy.
Reporting side effects
You can help ensure medicines are safe by reporting the side effects you experience.
You can report side effects to your doctor, or directly at www.tga.gov.au/reporting-problems
Brand name
(ARTG)
: RITALIN LA methylphenidate hydrochloride 10 mg modified release capsule bottleConsumer Medicine Information (CMI)
Read the CMI leaflet for facts you need to know before, during and after taking your medicine.
For more information about CMIs and how to read them, please visit How to read Consumer Medicine Information (CMI).
What this medicine is used for
(ARTG)
Ritalin 10 tablets and Ritalin LA capsules are indicated for the treatment of ADHD. Ritalin 10 tablets are also indicated for the treatment of narcolepsy. Attention-Deficit Hyperactivity Disorder (ADHD) ADHD was previously known as attention-deficit disorder. Other terms used to describe this behavioural syndrome include: minimal brain dysfunction in children, hyperkinetic child syndrome, minimal brain damage, minimal cerebral dysfunction, minor cerebral dysfunction and psycho-organic syndrome of children. Ritalin 10 / Ritalin LA are indicatedas an integral part of a total treatment program for ADHD that may include other measures (psychological, educational and social) for patients with this syndrome. Stimulants are not intended for use in the patient who exhibits symptoms secondary to environmental factors and/or other primary psychiatric disorders, including psychosis. Special diagnostic considerations for ADHD in children: The aetiology of this syndrome is unknown and there is no single diagnostic test. Adequate diagnosis requires the use, not only of medical, but also of psychological, educational and social resources. Characteristics commonly reported include: chronic history of short attention span, distractibility, emotional lability, impulsivity, moderate to severe hyperactivity, minor neurological signs and an abnormal EEG. Learning may or may not be impaired. The diagnosis must be based upon a complete history and evaluation of the child and not solely on the presence of one or more of these characteristics. Drug treatment is not indicated for all children with this syndrome. Stimulants are not intended for use in children who exhibit symptoms secondary to environmental factors (e.g. child abuse in particular) or primary psychiatric disorders. Appropriate educational placement is essential and psychosocial intervention is generally necessary. When remedial measures alone are insufficient, the decision to prescribe stimulant medicine will depend upon the physician's assessment of the chronicity and severity of the child's symptoms. Continuation of treatment in adolescent and special diagnostic considerations for ADHD in adults: There is limited information to guide clinicians about how long older adolescents should continue to receive treatment with drugs for attention deficit hyperactivity disorder (ADHD). The decision should be based on the extent to which symptoms of ADHD and social functioning have improved to a point that medication is no longer needed. If older adolescents have been largely symptom-free for a year and are functioning well, a trial without medication is warranted. This should be undertaken at times of low stress such as during holidays or in a period when a school routine is well established. ADHD needs to be considered in adults who present with longstanding symptoms suggestive of ADHD (inattention, impulsivity, disorganisation) that appear to have started in childhood and are persisting into adult life. Further, people with personality disorder and/or problems with drug use accompanied by a significant level of impulsivity and inattention should be referred for evaluation by a psychiatrist with the training and skills required to assess and treat ADHD. This expertise is necessary due to the overlap of ADHD symptoms with anxiety, mood and personality disorders. Narcolepsy The symptoms include daytime sleepiness, inappropriate sleep episodes and rapidly occurring loss of voluntary muscle tone. Ritalin 10 is effective for symptoms of sleepiness but not for loss of voluntary muscle tone.
How to use this medicine
(ARTG)
This medicine contains one component only.
Component :
- Capsule, modified release
- Oral
- White to off-white beads in light brown opaque hard gelatin capsule with "NVR" on cap and "R10" on a white opaque body in tan ink.
Images

Images © Medicines Information Pty Ltd.
Storage conditions
(ARTG)
- Store below 30 degrees Celsius
- Keep Container Tightly Closed/Airtight
- Shelf lifetime is 24 Months.
Do I need a prescription ?
(ARTG)
These medicines require authorisation for prescription from your doctor. It is Schedule 8 : Controlled Drug.
- 100 pack
- 30 pack
Is this medicine subsidised ?
(PBS)
This medicine was verified as being available on the PBS (Pharmaceutical Benefits Scheme) on May, 1 2026. To learn more about this subsidy, visit the Pharmaceutical Benefits Scheme (PBS) website.
Pregnant or planning a pregnancy ?
(AHT)
For the active ingredient methylphenidate
You should seek advice from your doctor or pharmacist about taking this medicine. They can help you balance the risks and the benefits of this medicine during pregnancy.
Reporting side effects
You can help ensure medicines are safe by reporting the side effects you experience.
You can report side effects to your doctor, or directly at www.tga.gov.au/reporting-problems









